
A sourd dough bread made from a spontaneous starter
After 7 days of feeding my sour dough starter “took off” and was ready for baking. Even with a water bath set to 28 °C it took longer than expected. 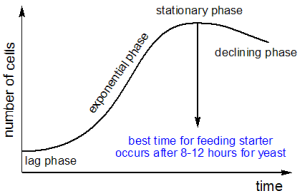 I started off with 100% hydration as this is convenient when you have to feed your starter frequently. Using only whole grain rye flour and water, I fed my starter every 12 hours (I’ve included details of the “feeding schedule” at the end of this post). This time interval is based on the growth cycle of yeast, where the yeast after an exponential growth phase reaches a plateau after 8-12 hours. This is the best time for feeding the starter.
I started off with 100% hydration as this is convenient when you have to feed your starter frequently. Using only whole grain rye flour and water, I fed my starter every 12 hours (I’ve included details of the “feeding schedule” at the end of this post). This time interval is based on the growth cycle of yeast, where the yeast after an exponential growth phase reaches a plateau after 8-12 hours. This is the best time for feeding the starter.
There seems to be a consensus that a wet starter (i.e. 100% hydration) favors growth of lactic acid bacteria (LAB) which in turn produce acids. The low pH after 2 days in my starter suggests plenty of LAB activity, so the main challenge for me was to get the yeast growing. Considering the fact that the yeasts found in sourdoughs prefer areob conditions for growth, I should have whisked in more air with each addition of water. And I wonder if this is the origin of the widespread myth that you “catch wild yeasts from the air”. I’m quite sure whisking helps, but what you do is not to catch yeast, but rather feed oxygen to your starter. This needs testing though! Most starter recipes call for discarding of half or even more of the starter before each feeding. Even though it seems wasteful I wonder if an important effect of this is to dilute the acid produced by the LAB (the flour may also act as a buffer). This acid will to some extent slow the growth of yeasts (even though the sourdough yeasts are far more acid tolerant than the conventional brewer/baker’s yeast Saccharomyces cerevisia).

Bubbles indicating that the starter is active
Regarding temperature the growth optimum for LAB lies around 32-33 °C whereas the growth optimum for yeast is somewhat lower at 28 °C. The sourdough FAQ has further temperature recommendations which are illustarted in the graph below (data from sourdough FAQ). Holding this together with the notion (see for instance p. 272 in “The handbook of dough fermentations”) that lower temperatures (20-25 °C) favor acetic acid production (= stronger flavor) and higher temperature (> 32 °C) favors lactic acid production (= milder flavor) it immediately becomes clear why a starter kept at roomtemperature has little yeast activity and smells of acetic acid. It need not be ruined, but is desperately in need of dilution, aeration and higher temperature.
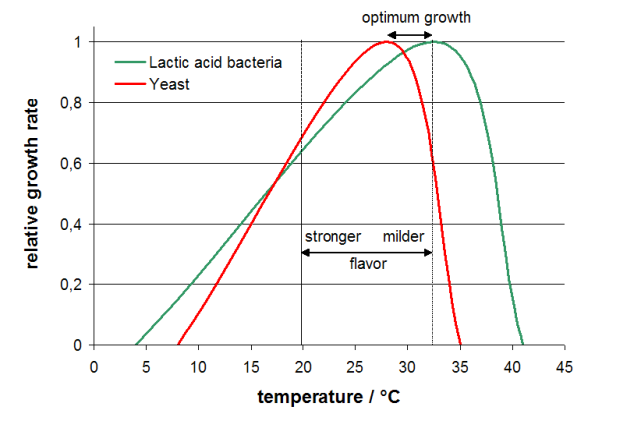
Effect of temperature on growth of lactic acid bacteria and yeast based on data for optimum growth and no growth from the sourdough FAQ
I’ve kept the starter alive since August and baked with it at least once a week. I make sure that I keep 50-100 g which I store in the fridge. I’ve also frozen a sample just in case. I’ve changed from 100% hydration to 67% hydration, as this simplifies the calculations a little. On the evening before baking day I feed the starter to a total starter weight of approximately 900 g. The next day I bake bread as follows (the exact numbers were calculated using an internet sour dough calculator with the following input: 3200 g total dough weight, 25% starter, 67% hydration of starter and final dough and 1.8% salt):
Sour dough bread
949 g water
1417 g flour *
800 g starter (67% hydration, 25% of total dough)
34 g salt
* for instance 300 g rye whole grain, 200 g rye fine, 300 g whole grain wheat and then plain all-purpose wheat flour up to 1417 g.
Mix water and flour mixed until all flour is wetted. Leave for 15-20 minutes (during which the autolyse proceeds – this eases subsequent mixing). Mix (see more comments below regarding method/machine for this) until dough is smooth, and while mixer is running add sour dough starter. Once the starter has been properly incorporated into the dough, add the salt. Cover and leave to rise until volume has increased 30-100% (I know – this is not very accurate…). I have left it in on my bench top, but while this worked well on warm August days, it seems to be less than ideal on colder October days. Leaving the dough to rise on top of the fridge might be a conventient compromise here as my current waterbath is not large enough to hold the mixing bowl with dough. The reason I use a starter with a 67% hydration is that I can be more sloppy when adding the starter to the sourdough as it will not change the hydration of the dough. After proofing and slashing I bake the breads on a baking stone which is preheated to 250 °C. Right beneath the baking stone I have a small oven proof dish that I fill with boiling water. This helps to moisten the air in the oven and it simulates the steam injection port of professional baking ovens. There are several reasons why this is important. Moist air is a better heat conductor than dry air, and it prevents the surface from drying out too early while baking, resulting in a better oven spring. Furthermore the moist air condenses on the cold surface of the dough which improves gelatinization of the starch. This in turn gives better crust formation. After 10 min at 250 °C I open the oven to let the moisture out, take out the dish with water, turn the heat down to 220 °C and close the oven door. I bake the breads to a core temperature of about 93-95 °C.

Here I bake two 800 g loaves on a baking stone. Notice the dish with water for steam generation.
I should comment on mixing. When kneading by hand I’ve had a tendency to add to much flour. In fact I think this is one of the reasons why I quit baking bread several years ago – I found that the breads I made generally were a little to dry with a poor crumb, and at that time I didn’t really sit down and think about these matters. In retrospect however there’s no doubt that baking bread and adding flour ad lib until the dough feels good to touch is NOT recommended. Well anayway not unless you stop adding flour while the dough is still quite sticky. The thing about doughs with a high percentage rye is that they are quite sticky, and they should be. This is the best argument you’ll ever get for buying a kitchen gadget: bread doughs are too sticky to be kneaded by hand! There you have it! I’ve settled with the Assistent from Sweden. It was formerly sold under the Electrolux brand, but is now marketed indepentendly, yet it is still produced at the very same factory as always. In the US the machine is known as the Magic Mill. It has a huge 7 L bowl that rotates. The roller is attached to a flexible arm, so if the dough is to hard to work the arm just moves to the middle of the bowl. This significantly reduces the chances of overheating the motor. There are of course other alternatives from Kitchen Aid, Hamilton Beach and Viking Range which seem robust, but I have no experience with these. However, I doubt that the average Kenwood can cope with more than 3 kg of bread dough (but please correct me if I’m wrong :)).

Unlike most other machines, the bowl of the Magic Mill/Assitent rotates while the roller pushes the dough to the sides of the bowl
Sources for further reading
I’ve read quite a bit about sourdoughs, and what I’ve been looking for a simple correlation between temperature, hydration and fermentation time. An excellent source of information with lots of practical advice is the FAQ from rec.food.sourdough.
I think the best resource I’ve found sofar is Lorenz and Bruemmer’s chapter “Preferments and Sourdoughs for German Breads” and Teija-Tuula Valjakka, Heikki Kerojoki and Kati Katina’s chapter “Sourdough Bread in Finland and Eastern Europe” in “The handbook of dough fermentations”. I will have to study these more carefuly.
There are quite a number of academic publications which also touch upon the effect of temperature on acid development. Here are some snippets:
“Controlled production of acetic acid in wheat sour doughs”:
…temperature has no significant effect, and that fructose is more efficient in influencing the FQ than dough yield.
FQ = fermentation quotient = lactic acid / acetic acid
… Low temperature (25 degrees C) and sour dough firmness (dough yield 135) were appropriate for LAB souring activities but limited yeast metabolism. Raising the temperature to 30 degrees C and semi-fluid sour doughs gave more complete volatile profiles …
“Sourdough: a tool for the improved flavour, texture and shelf-life of wheat bread” (Ph.D. thesis of Kati Katina):
The production of acids depends also on other things such as fermentation temperature, time and dough yield. Optimum temperatures for the growth of lactobacilli are 30-40 °C depending on strain (Stanier et al. 1987) and for yeasts 25-27 °C. In general, a higher temperature, a higher water content of sourdough and the utilisation of wholemeal flour enhances the production of acids in wheat sourdoughs (Brummer and Lorenz 1991, Lorenz and Brummer 2003).
I must admit that I’m a little confused as some of these snippets seem to contradict. It might be that I’m overlooking something important though and that I’m taking results out of their context. Any insight from my readers on this will be greatly appreciated!
Starter details
Here’s the details from my notebook on how I fed my sourdough starter. As I mentioned above, I wonder if discarding dough in the process actually does make sense after all.
- July 29, evening: 50 g rye + 50 g water
- July 30, morning: 25 g rye + 25 g water, evening: small bubbles (!), 50 g rye + 50 g water
- July 31, morning: 50 g rye + 50 g water, evening: pH measured to 3-4 with strips, fed with 50 g rye + 50 g water
- August 1: morning: 50 g rye + 50 g water, tested for bread baking, result: not active enough, feeding continued evening: 50 g rye + 50 g water
- August 2: morning: 50 g rye + 50 g water, evening 50 g rye + 50 g water
- August 3: morning: 50 g rye + 50 g water, evening: no bubbles, discarded all except ~100 g, fed with 50 g rye + 50 g water
- August 4: morning: 50 g rye + 50 g water, evening 50 g rye + 50 g water
- August 5: morning: 50 g rye + 50 g water, evening: big bubbles, the starter is active, CO2 production evidenced by tickling in nose, fed with 60 g rye + 60 g water and 2 x 100 g samples taken for fridge and freezer as fallback points
- August 6: morning: 50 g rye + 50 g water, first successfull bread made with the starter
I show my raw data to illustrate that it’s not straightforward, even with temperature control.
A fool proof starter – is it possible?
What I’m hoping to achieve can be summarized as follows: A “fool proof”, robust and quick method to obtain a sourdough starter that’s as simple as possible, using only flour and water (possibly with addition of some fruit) without having to waste anything of the starter. Temperature is maintained using a thermostated water bath. Preferably it should be possible to adjust the fermentation quotient (ratio of lactic acid/acetic acid) and the total titrable acid content by means of temperature, time and hydration/dough yield.

I think when people talk about yeasts in sourdough, they often times mean both yeast and lactic acid bacteria. Lay people don’t usually understand the difference. Using that definition, you can indeed catch them from the air. I have started many cultures from scratch (not using starter). Granted, I didn’t use sterile flour, but bacteria and yeast are everywhere. I think a lot of the old recipes for beer used wild yeasts, bock for one, if i recall correctly.
I keep my sourdough in the fridge, and feed it sporadically. It’s never failed me. Though now it tends to smell like bananas sometimes. I’d love to make it so it would do that consistently, but I think that would take a tiny bit of genetic engineering.
Hi my name is alex and i am a chef living in las vegas in the us i was wondering where you bought your Magic Mill i so far only found it on your amazon link and was wondering what store you bought yours at. At 27 lb shipping is not a thing i want to pay.
Thank you
Alex Schubert
Alex: Living in Norway I got my from a Norwegian internet shop – I guess it won’t help you much in the US.
At the risk of introducing an irrelevant detail — so how does your bread *taste*???? (-:
Martin – very thorough, most helpful and enjoyable as usual but with one vital piece missing…
How do they TASTE?
The yeasts, when in an aeróbic media multiply themselves, and in an anaeróbic media they produce the methabolites ethanol and CO2. That is the reazon for whipping.
Good work, very enlightening. A couple of comments/questions:
– dilution of acid vs culture: when diluting, the yeast concentration will also be diluted, ..or? Would this make a real difference? Might it be that more flour is needed simply to feed the yeast (i.e. dough is depleted of maltose?)
– If the four acts as a buffer, what components in the flour might do so?
– Optimum growth temperature: an interesting case of something that might seem counterintuitive. I guess this counters the (my) general notion in cooking that higher temperature might result in stronger flavour. A potential case of a culinary precision, which I guess there might be a host of in baking
The aroma is pretty close to a traditional German “Bauernbrot”. The crumb is soft, yet it slices very well so you can easily obtain thin slices without everything falling apart. The bread also keeps well, i.e. it doesn’t get to hard/dry upon storage.
Regarding dilution: I’ve also been pondering on this – could it be that yeasts multiply faster than the LAB, but that the LAB are so effective at producing acid that it halts the yeast multiplication. If this is the case, for every dilution the yeast/LAB ratio should change in favor of yeast. Does this reasoning make sense?
re: dilution
I am researching the science behind sourdough fermentation. So call me sourdough experience stupid right now.
If I understand what you are questioning, this is what I found:
My guess would be controlling PH.
“Both the yeast and lactobacilli are inhibited by the acid produced” The yeast will die eventually.
Read more: http://www.faqs.org/faqs/food/sourdough/faq/preamble.html#ixzz0Uyb4Ngcx
re: Bananas
Being a beer brewer I have a guess on the banana smell. In brewing beer an ester can be a desirable thing giving a certain beer a positive character.
It could be an ‘ester’ formation that comes from some kind of acid reacting to alcohol.
expanding my guess, storing in the frig for long periods and not refreshing more often could be allowing these esters to form.
I have not made sourdough yet as I have to need to learn the science before I bake something like this, I liken the development or fermentation of the dough to making home made cultured buttermilk, which I have made a lot of. The fermentation method is identical. In with some of the old (growing culture) to make way for some new. It was explained to me by a professor that what you are doing with making cultured buttermilk (lactic acid) is ‘purifying’ the culture. Going from simple sour milk to something drinkable.
This is what I have come to conclude so far with fermenting dough to make sourdough. It goes through a purification process. If you leave the buttermilk culture in the fridge long enough, it goes bad. Where the difference would be, yeast makes alcohol that known to a brewer when mixed with certain acids, forms esters – smells and flavors.
Great article by the way…
first sourdough out of oven and vry proud!!! little more salt and maybe extra 10min!! but hey.. i followed a link and found a milk/flour starter, sitting in my warm furnace room over a week with ur feeding plan and tips produced a smooth airy starter with a lovely yeasty smell. bread is full of those lovely air pockets and by adding 55g wack of malt gave me beautiful crisp crust… cheers
also slowly gettin thru ur hydrocolloid recipe.. ups and downs but fun all the way.
awsome awsome site, kep itn up
cheers willl get some photos and be in touch for tips
lee arhus, dk